In a groundbreaking study published February 9, 2026, in the Proceedings of the National Academy of Sciences (PNAS), researchers at Washington University School of Medicine in St. Louis—partnered with the VA St. Louis Health Care System—have demonstrated that engineered immune cells can effectively target and reduce the toxic amyloid beta plaques driving Alzheimer’s disease. The findings, derived from mouse models, mark the first time CAR-T cell therapy, a cutting-edge technique originally developed to combat blood cancers, has been successfully adapted to treat a neurodegenerative condition. For the Department of Veterans Affairs, where Alzheimer’s and related dementias already affect 10% of patients aged 65 and older and are projected to surge by 22% over the next decade, the research offers a critical glimpse into potential future therapies amid a growing crisis.
Why Veterans Face a Unique and Growing Dementia Crisis
The Department of Veterans Affairs is confronting a dementia epidemic that is both urgent and uniquely tied to military service. According to the Veterans Health Administration, approximately 276,000 VA patients aged 65 and older were living with dementia in 2020. By 2033, that number is projected to climb to 335,000—a 22% increase in just over a decade. This demographic shift is particularly alarming because more than half of all VA patients are now 65 or older, placing them squarely in the high-risk window for neurodegenerative diseases. The crisis is compounded by the fact that veterans face elevated risks for dementia that civilians do not. Large-scale longitudinal studies have shown that veterans with post-traumatic stress disorder (PTSD) are 50% to 60% more likely to develop dementia later in life. The connection between traumatic brain injury (TBI) and cognitive decline is even more pronounced. A January 2026 study published in Frontiers in Dementia found that veterans carry a higher aggregate prevalence of nearly every modifiable dementia risk factor, including diabetes, chronic pain, smoking, depression, sleep disturbances, and hearing loss.
The Intersection of Service-Connected Conditions and Alzheimer’s Risk
Research from the VA’s Million Veteran Program has illuminated a troubling synergy between military service and genetic vulnerability to Alzheimer’s. The program’s data reveals that PTSD and TBI not only increase dementia risk on their own but also interact with genetic predispositions to the disease. This means that veterans who served in recent conflicts—particularly post-9/11 troops—may face compounded biological vulnerabilities as they age. Dr. Jonathan Kipnis, the neuroimmunologist leading the Washington University study, has emphasized that this ‘coming wave’ of dementia among veterans is not a hypothetical future concern but a current and escalating reality. The VA is already bracing for the impact, with limited treatment options available for a patient population that has historically borne the brunt of service-related health complications.
How CAR-T Cell Therapy Could Revolutionize Alzheimer’s Treatment
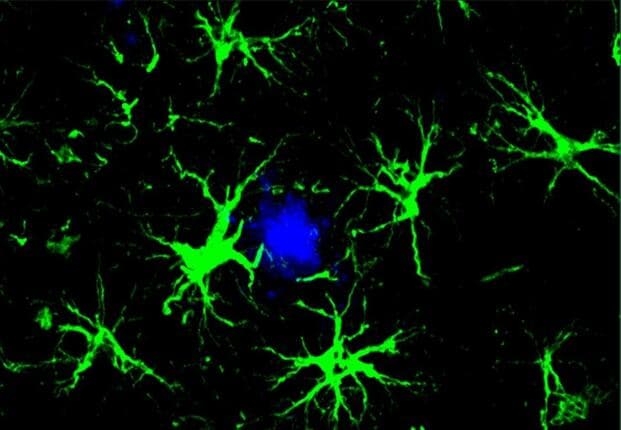
CAR-T cell therapy—short for chimeric antigen receptor T-cell therapy—has transformed the treatment of certain blood cancers over the past decade. The technique involves extracting a patient’s own T cells, genetically engineering them to recognize and attack a specific target, and then reinfusing them into the body. In the Alzheimer’s study, researchers led by Dr. Kipnis applied this principle to target amyloid beta, the sticky protein aggregates that form plaques in the brains of Alzheimer’s patients and are widely considered a key driver of the disease. The team removed T cells from healthy mice, modified them to seek out amyloid beta, and administered three injections spaced 10 days apart to six-month-old mice genetically engineered to develop Alzheimer’s-like plaques. The results were striking: treated mice showed a significant reduction in amyloid beta plaques compared to controls, alongside diminished activation of microglia and astrocytes—cells linked to the neuroinflammation that accelerates cognitive decline.
From Mice to Humans: The Long Road Ahead
Despite the promising findings, the study’s authors caution that the research is still in its infancy. The experiments were conducted exclusively in mice, and while the results suggest a potential pathway for human treatment, numerous hurdles remain. Key questions include determining safe and effective dosing, understanding long-term effects, and assessing whether the approach can translate to the far more complex architecture of the human brain. Dr. Kipnis has noted that the team’s next steps involve exploring whether the engineered cells can improve broader brain health beyond plaque reduction, as well as testing the therapy in mouse models of other neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS) and Parkinson’s disease. The timeline for human trials is uncertain, but the research builds on a decade of Kipnis’s groundbreaking work, including his 2015 discovery of the brain’s meningeal lymphatic system and his recent findings that neuroprotective T cells can aid spinal cord recovery.
The VA’s Role in Pioneering Neurodegenerative Disease Research
The collaboration between Washington University and the VA St. Louis Health Care System exemplifies the federal government’s commitment to addressing the intersection of military service and neurological health. The VA has long been a leader in research on TBI, PTSD, and their long-term consequences, with initiatives like the Million Veteran Program providing critical data on genetic and environmental risk factors for dementia. The St. Louis partnership is part of a broader ecosystem of VA-funded research centers across the country, where scientists are exploring innovative therapies for conditions that disproportionately affect veterans. For example, the VA’s National Center for PTSD has documented how chronic stress from military service can accelerate brain aging, while VA hospitals nationwide are piloting early detection programs for cognitive decline using advanced imaging and biomarker analysis. The CAR-T study, while preliminary, underscores the VA’s growing focus on leveraging immunotherapies—traditionally used in oncology—to tackle neurodegenerative diseases.
Key Takeaways: What Veterans and Families Need to Know
- CAR-T cell therapy, a cancer-fighting technique, has been adapted to reduce toxic amyloid beta plaques in mouse models of Alzheimer’s—a first in neurodegenerative disease research.
- Veterans face a 22% projected increase in dementia cases by 2033, with service-connected conditions like PTSD and TBI compounding genetic risks for Alzheimer’s.
- The study, led by Dr. Jonathan Kipnis at Washington University and the VA St. Louis Health Care System, shows promise but is still years away from human trials.
- Broader implications of the research include potential applications for other neurodegenerative diseases like ALS and Parkinson’s, as well as insights into the brain’s immune response.
- The VA is investing in early detection and treatment innovation, positioning itself at the forefront of neurodegenerative disease research for veterans.
A Glimpse into the Future of Alzheimer’s Care for Veterans
While the CAR-T breakthrough offers hope, the reality for veterans and their families remains sobering. The average age of dementia onset is 65, and many post-9/11 veterans are now entering this critical window. The VA’s current dementia care model relies heavily on supportive services such as memory care programs, caregiver support, and early intervention strategies, but effective disease-modifying treatments are scarce. The Washington University-VA study suggests that immunotherapies could one day fill this gap, but significant challenges lie ahead. Regulatory hurdles, ethical considerations, and the sheer complexity of the human brain mean that even optimistic projections place human trials years away. In the meantime, the VA is ramping up efforts to identify at-risk veterans through programs like the Mild Cognitive Impairment (MCI) screening initiative and is expanding access to clinical trials for emerging therapies, including anti-amyloid drugs and lifestyle intervention studies.
Expert Perspectives: The Promise and Pitfalls of Immunotherapy for Alzheimer’s
This study represents a paradigm shift in how we think about treating Alzheimer’s—not just as a disease of abnormal protein accumulation, but as a condition where the immune system can be harnessed for therapeutic benefit. The fact that we can engineer T cells to target specific pathological features in the brain opens up entirely new avenues for intervention. That said, we are still in the very early stages. Translating this from mice to humans will require rigorous safety testing, careful dose optimization, and a deep understanding of how these cells interact with the broader immune environment in the brain.
For veterans, the stakes could not be higher. The convergence of PTSD, TBI, and aging is creating a perfect storm of cognitive decline risk. If therapies like CAR-T can be safely developed, they could transform not just the lives of individual veterans, but the entire landscape of dementia care within the VA system.
What’s Next? The Road from Lab to Clinic
The Washington University-VA team is not alone in exploring immunotherapies for Alzheimer’s. Dozens of research groups worldwide are investigating various approaches, from monoclonal antibodies that clear amyloid plaques to vaccines designed to stimulate the immune system. What sets the CAR-T work apart is its precision—engineered T cells can actively seek out and destroy pathological proteins while potentially sparing healthy tissue. However, the approach also carries risks, including cytokine release syndrome (a severe inflammatory response) and the possibility of unintended immune reactions. The next phase of research will focus on refining the therapy’s safety profile, testing it in larger animal models, and addressing logistical challenges such as the need for personalized cell engineering. Dr. Kipnis has indicated that his team will next explore the therapy’s effects in mouse models of frontotemporal dementia and chronic traumatic encephalopathy (CTE), conditions that share pathological features with Alzheimer’s and are highly relevant to the veteran population.
How Veterans Can Stay Informed and Advocate for Progress
For veterans concerned about their cognitive health or that of a loved one, staying informed about ongoing research and clinical trials is crucial. The VA’s Alzheimer’s Disease Research Centers of Excellence, located in 29 academic medical centers across the country, offer cutting-edge care and opportunities to participate in studies. Additionally, organizations like the Alzheimer’s Association and the Wounded Warrior Project provide resources, support groups, and advocacy tools to help veterans navigate the complex landscape of dementia care. Veterans can also access the VA’s Geriatric and Extended Care services, which include cognitive screening and early intervention programs. As research advances, advocates are pushing for increased funding and streamlined pathways to clinical trials, particularly for conditions linked to military service.
Frequently Asked Questions
Frequently Asked Questions
- What is CAR-T cell therapy, and how does it work for Alzheimer’s?
- CAR-T therapy involves genetically modifying a patient’s T cells to recognize and attack specific targets—in this case, amyloid beta plaques in the brain. The modified cells are then reinfused to seek out and destroy pathological proteins associated with Alzheimer’s disease.
- Why are veterans at higher risk for Alzheimer’s and dementia?
- Veterans face elevated risks due to service-connected conditions like PTSD and TBI, which increase dementia risk by 50% to 60% or more. Additionally, veterans have higher rates of modifiable risk factors such as diabetes, chronic pain, and depression, all of which contribute to cognitive decline.
- When will CAR-T therapy for Alzheimer’s be available to veterans?
- While the mouse study is promising, human trials are likely years away. Researchers must first address safety, dosing, and efficacy in larger animal models before progressing to clinical trials. The VA is actively investing in early detection and treatment innovation to prepare for future breakthroughs.