In a groundbreaking advancement for cancer immunotherapy, researchers have successfully reprogrammed human immune cells directly inside living organisms to fight tumors—a process known as in vivo CAR T cell engineering that bypasses the costly and time-consuming ex vivo manufacturing currently required for these lifesaving treatments. Using a sophisticated CRISPR-Cas9 system combined with adeno-associated viral vectors, the team targeted the endogenous T-cell receptor alpha locus (TRAC) to integrate therapeutic chimeric antigen receptor (CAR) sequences into primary human T cells while they remain inside a living host. Published in the peer-reviewed journal Nature, this study represents the first demonstration of site-specific integration of large DNA payloads in human T cells in vivo, achieving therapeutic levels of cancer-fighting CAR T cells that completely eliminated aggressive tumors in multiple humanized mouse models, including B-cell acute lymphoblastic leukemia, multiple myeloma, and sarcoma.
- Scientists developed the first in vivo method to generate therapeutic CAR T cells inside living organisms, eliminating the need for ex vivo manufacturing
- The CRISPR-AAV approach targets the TRAC locus to ensure T cell-specific CAR expression and prevent off-target effects
- In humanized mouse models, the technique achieved 100% tumor control in leukemia and complete responses in myeloma and sarcoma
- The engineered vectors were optimized to resist human neutralizing antibodies and specifically target T cells, overcoming major delivery barriers
- This breakthrough could enable off-the-shelf CAR T therapies from universal donors, dramatically reducing costs and wait times
Revolutionizing Cancer Therapy: From Factory-Made to Body-Made CAR T Cells
Chimeric antigen receptor T-cell therapy has transformed the treatment landscape for certain blood cancers, with seven FDA-approved products now available for patients with hematologic malignancies. However, the current standard-of-care approach requires extracting a patient's T cells through leukapheresis, shipping them to specialized manufacturing facilities for genetic modification, and then reinfusing the engineered cells—a process that typically takes 2-4 weeks, costs hundreds of thousands of dollars, and produces variable product quality depending on the patient's starting T-cell population. These limitations have spurred intense research into allogeneic approaches using T cells from healthy donors, creating "off-the-shelf" products that could be administered immediately to any patient. While clinical trials of allogeneic CAR T cells have shown promise, the cells eventually get rejected by the patient's immune system, leading to relapse in many cases.
The Critical Need for In Vivo Engineering
The fundamental challenge with current CAR T manufacturing is the need to remove T cells from the body, modify them in artificial environments, and then return them—an inherently inefficient and expensive process. Direct in vivo engineering addresses these limitations by performing the genetic modification while T cells remain in their natural microenvironment, potentially preserving their native functionality and avoiding the activation-induced exhaustion that often occurs during ex vivo manufacturing. Prior attempts at in vivo CAR T cell generation have used randomly integrating viral vectors for constant CAR expression or lipid nanoparticles that provide only temporary expression, both of which face significant hurdles in achieving therapeutic doses and T cell specificity. The risk of off-target effects is substantial: engineering hematopoietic stem cells could lead to transformational mutagenesis, while CAR expression in tumor cells could prevent antigen recognition and cause treatment-resistant relapse.
The CRISPR-AAV Breakthrough: Targeting the TRAC Locus for Precise Genetic Integration
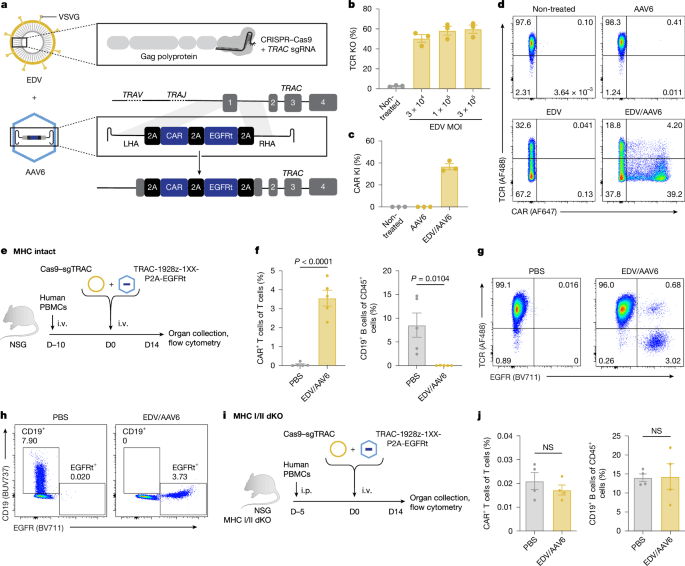
The research team developed a two-vector system combining enveloped delivery vehicles (EDVs) for CRISPR-Cas9 ribonucleoprotein delivery with adeno-associated virus serotype 6 (AAV6) for homology-directed repair template (HDRT) delivery. By targeting the TRAC locus—the gene encoding the T-cell receptor alpha chain—they ensured that CAR expression would be driven by the endogenous TRAC promoter, which is exclusively active in T cells. This approach eliminates the need for external promoters that could drive CAR expression in non-T cells, addressing one of the major safety concerns in gene therapy. The TRAC targeting also disrupts the native T-cell receptor, a necessary step to prevent graft-versus-host disease in allogeneic settings where CAR T cells from healthy donors are used.
Overcoming Three Critical Barriers to In Vivo Delivery
The researchers identified three major obstacles to successful in vivo engineering: susceptibility of AAV vectors to neutralizing antibodies in human blood, lack of specificity for T lymphocytes, and limited availability of cycling T cells for efficient gene editing. To address these challenges, they employed a sophisticated evolutionary strategy to create a new AAV variant called AAV-hT7, which showed approximately 20,000-fold enrichment from the parental AAV6 library after three rounds of selection on human T cells in the presence of human serum. This evolved capsid achieved efficient transduction even in the presence of neutralizing antibodies that would normally block AAV6.
The team then enhanced delivery specificity by engineering the EDV envelope glycoprotein to incorporate an anti-CD3 single-chain variable fragment, which both targets the CD3 component of the T-cell receptor complex and induces T-cell activation. This dual-purpose modification ensures that only T cells receive the gene editing payload while simultaneously promoting their activation—essential for the cell cycling required for efficient homology-directed repair. The combination of AAV-hT7 and anti-CD3-EDV created a system with unprecedented specificity, showing minimal gene integration in non-T cells including hematopoietic stem cells, natural killer cells, macrophages, and even cancer cell lines.
From Petri Dish to Living Organism: Proving Efficacy in Humanized Mouse Models
The proof-of-concept experiments were conducted in humanized mouse models created by engrafting immunodeficient NSG mice with human peripheral blood mononuclear cells. These "humanized" mice represent the closest approximation to human physiology available in preclinical research. When treated with the dual EDV/AAV system, the mice generated therapeutic levels of TRAC-CAR T cells that expanded to comprise up to 19.7% of all splenic T cells within just two weeks. More significantly, these in vivo engineered cells achieved complete B-cell aplasia in the treated mice, indicating functional CAR activity. Spectral flow cytometry analysis revealed that the engineered T cells maintained memory markers and displayed a progenitor exhausted phenotype associated with improved antitumor activity.
Tumor Control Across Multiple Cancer Types
The therapeutic efficacy of in vivo engineered CAR T cells was demonstrated across three distinct tumor models. In B-cell acute lymphoblastic leukemia (B-ALL) models using the NALM6 cell line, 18 out of 20 mice across four different human donors achieved complete responses after a single treatment. When rechallenged with tumor cells 39 days after initial treatment, these mice maintained durable tumor control, demonstrating functional persistence of the engineered cells. Similar success was achieved in multiple myeloma models using the OPM2 cell line, where all 8 treated mice achieved complete responses, with 3 out of 4 maintaining control after tumor rechallenge. Perhaps most impressively, the technology extended to solid tumors in sarcoma models, where 5 out of 6 mice achieved complete responses, marking the first demonstration of in vivo CAR T cell generation against solid malignancies.
Why This Approach Could Transform CAR T Therapy: Advantages Over Current Methods
The in vivo engineering approach offers several compelling advantages over traditional ex vivo manufacturing. First, it eliminates the weeks-long delay between T-cell extraction and reinfusion, allowing for immediate treatment of critically ill patients. Second, it creates more uniform CAR expression across the engineered cell population since every successfully edited T cell expresses the CAR under control of the endogenous TRAC promoter, rather than relying on variable viral integration sites. Third, it significantly reduces manufacturing complexity and cost by avoiding the need for specialized cell processing facilities. Most importantly, it enables the creation of off-the-shelf CAR T products from universal donors, which could be banked and administered to any patient without concern about immune rejection.
Comparing In Vivo Engineered Cells to Traditional CAR T Products
When benchmarked against both lentiviral CAR T cells and ex vivo manufactured TRAC-CAR T cells, the in vivo engineered cells demonstrated superior performance. The engineered cells expanded more rapidly, peaking at week 2 compared to week 3 for lentiviral products, and achieved 8-50 times greater expansion at peak levels. They maintained higher and more consistent CAR expression across the cell population, avoiding the heterogeneous expression patterns that plague lentiviral transduction. The progenitor exhausted phenotype observed in the engineered cells aligns with recent findings that these cells demonstrate improved persistence and antitumor activity in multiple tumor models.
Safety Considerations and Addressing Potential Risks
While the promise of in vivo engineering is substantial, safety remains paramount. The researchers carefully evaluated several potential risks. First, they confirmed that the engineered vectors did not induce systemic inflammation or cytokine release syndrome, with cytokine profiling showing no significant elevation at 1 and 7 days post-treatment. Second, they demonstrated that the targeting specificity prevented CAR expression in tumor cells, which could otherwise lead to antigen-negative relapse through immune evasion. Third, the use of a promoterless CAR cassette ensures expression only occurs after successful integration at the TRAC locus, avoiding the constitutive expression that could drive CAR expression in packaging cells or other off-target sites.
Future Directions: From Mouse Models to Human Patients
The successful demonstration in multiple mouse models sets the stage for translation to human clinical trials. The research team has identified several key next steps, including optimizing dosing regimens, evaluating long-term persistence and safety, and expanding the approach to additional cancer types and CAR targets. The technology could also be adapted for other immune cell types beyond T cells, including natural killer cells and macrophages. Most significantly, this approach could enable the development of universal donor CAR T products that would be available immediately for any patient, dramatically expanding access to this revolutionary cancer therapy.
The Bottom Line: A Paradigm Shift in Cancer Immunotherapy
This breakthrough represents more than just an incremental improvement in CAR T cell therapy—it signals a fundamental shift in how we approach cancer immunotherapy. By moving the genetic engineering process from factory floors to living organisms, researchers have overcome the most significant limitations of current CAR T therapy while opening new avenues for treating a wider range of cancers. The ability to generate therapeutic-grade CAR T cells directly inside patients could revolutionize cancer treatment, making advanced cellular therapies more accessible, affordable, and effective for the millions of patients worldwide who desperately need better treatment options.
Frequently Asked Questions About In Vivo CAR T Cell Engineering
Frequently Asked Questions
- How does in vivo CAR T cell engineering differ from traditional ex vivo methods?
- Traditional CAR T therapy requires removing T cells from the patient, shipping them to specialized facilities for genetic modification, and then reinfusing the engineered cells. In vivo engineering performs the genetic modification while T cells remain in the patient's body, eliminating weeks of manufacturing time and potentially preserving T cell functionality.
- What makes the TRAC locus an ideal target for CAR integration?
- The TRAC locus is exclusively active in T cells, ensuring that CAR expression is restricted to the intended cell type. Targeting this locus also disrupts the native T-cell receptor, which is necessary to prevent graft-versus-host disease in allogeneic settings where T cells from healthy donors are used.
- What safety concerns does this approach address compared to current CAR T therapies?
- The approach prevents CAR expression in non-T cells, including hematopoietic stem cells and tumor cells, which could lead to mutagenesis or antigen-negative relapse. The use of a promoterless CAR cassette ensures expression only occurs after successful integration at the TRAC locus, avoiding constitutive expression risks.