In a significant advance against one of medicine’s most persistent challenges, researchers have discovered a synthetic opioid compound that delivers powerful pain relief while minimizing the dangerous side effects that have fueled the ongoing opioid crisis. The breakthrough, published in the journal Nature, centers on N-desethyl-fluornitrazene (DFNZ), a novel μ-opioid receptor (MOR) superagonist derived from the nitazene class of synthetic opioids. Unlike traditional opioids such as morphine or fentanyl—which are notorious for causing respiratory depression, addiction, and overdose—DFNZ demonstrated exceptional analgesic efficacy in rodent models with markedly reduced adverse effects, including minimal respiratory suppression and negligible withdrawal symptoms. This discovery arrives at a critical juncture, as the United States continues to grapple with over 100,000 annual opioid-related deaths, according to the Centers for Disease Control and Prevention, and the global search for safer pain management solutions intensifies.
The Opioid Crisis: Why Safer Analgesics Are Critical
The opioid epidemic, which began in the 1990s with the overprescription of opioid analgesics, has evolved into a public health catastrophe exacerbated by the proliferation of potent synthetic opioids. Fentanyl, a Schedule II controlled substance, is 50 to 100 times more potent than morphine and has been linked to tens of thousands of overdose deaths annually. In recent years, a new class of synthetic opioids known as nitazenes—first synthesized in the 1950s as potential painkillers—has entered the illicit drug supply. Compounds like etonitazene and metonitazene are up to 1,000 times more potent than morphine, leading to extreme overdose risks. The U.S. Drug Enforcement Administration placed several nitazenes into Schedule I in 2022, recognizing their high abuse potential and lack of accepted medical use. Despite this regulatory action, nitazenes continue to circulate in the recreational drug market, underscoring the urgent need for safer therapeutic alternatives.
How DFNZ Differs from Traditional Opioids: Mechanism and Structure
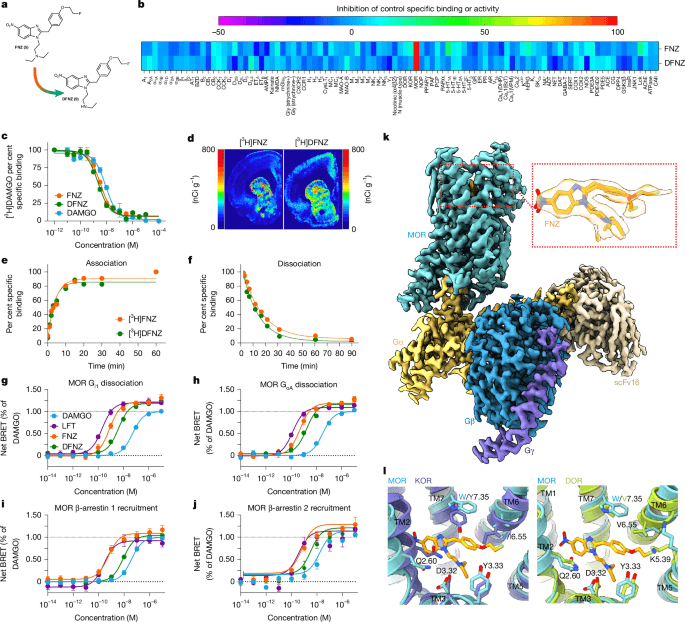
DFNZ belongs to a new generation of μ-opioid receptor agonists designed to maximize pain relief while minimizing harmful side effects. The compound is a metabolite of fluornitrazene (FNZ), a nitazene derivative synthesized by researchers to study MOR binding dynamics. FNZ exhibits ultra-high affinity for the μ-opioid receptor (Ki = 1.36 ± 0.11 nM) and activates the receptor with supramaximal efficacy—meaning it produces a stronger biological response than even the body’s natural opioid peptides. However, FNZ’s rapid metabolism in the liver produces DFNZ as its primary metabolite, which retains high MOR selectivity and potency (Ki = 1.00 ± 0.06 nM). The structural modifications in DFNZ—particularly the removal of an ethyl group during metabolism—alter its pharmacokinetic profile, reducing brain penetration and enhancing safety.
Nitazene Family: A Double-Edged Sword in Modern Medicine
Nitazenes were first developed in the 1950s by Swiss pharmaceutical company Hoffmann-La Roche as potential analgesic and anesthetic agents. Etonitazene, the prototype of this class, showed promise in preclinical trials but was abandoned due to its extreme potency and overdose risk. Today, nitazenes have resurfaced not as therapeutic agents, but as illicit drugs. Their presence in the recreational drug supply has been linked to numerous overdose deaths, prompting the DEA to classify several analogs as Schedule I substances. The structural versatility of nitazenes—allowing for modifications that enhance potency or alter metabolism—has made them both a scientific curiosity and a public health threat. DFNZ represents a paradigm shift: instead of exploiting nitazene toxicity, researchers have repurposed its molecular architecture to create a safer therapeutic.
From FNZ to DFNZ: A Metabolic Route to Safer Pain Relief
The journey from FNZ to DFNZ began with a focus on radiolabeling FNZ for positron emission tomography (PET) imaging studies. Researchers substituted a fluorine atom at the end of FNZ’s ethoxy chain to enable 18F labeling, a common technique in medical imaging. This modification inadvertently led to the discovery of DFNZ, which emerged as the primary metabolite of FNZ within 30 minutes of administration in mouse liver microsome studies. Unlike FNZ, which crosses the blood-brain barrier efficiently, DFNZ is actively effluxed by P-glycoprotein (PGP) and breast cancer resistance protein (BCRP), two primary transporters at the blood-brain barrier. This efflux mechanism limits DFNZ’s central nervous system penetration, reducing its potential for respiratory depression and addiction while preserving its analgesic efficacy.
In Vivo Evidence: DFNZ Delivers Pain Relief Without the Risks
In rodent models, DFNZ demonstrated potent analgesic effects in both acute and chronic pain scenarios. In a hot plate test assessing thermal nociception, DFNZ produced significant pain relief at doses approximately 60 times lower than morphine’s median effective dose (ED50 ≈ 0.3 mg/kg vs. morphine’s ~18 mg/kg). The compound also reversed mechanical hypersensitivity in a chronic inflammatory pain model induced by complete Freund’s adjuvant (CFA), restoring paw withdrawal thresholds to pre-injury levels. Crucially, DFNZ did not induce tolerance even after repeated daily administration, a common and devastating side effect of traditional opioids. Unlike morphine or fentanyl, which cause pronounced respiratory depression, DFNZ showed no significant decrease in brain oxygen levels at analgesic doses. In fact, at its maximal analgesic dose of 1 mg/kg, DFNZ induced a moderate but sustained increase in brain oxygenation—a stark contrast to the hypoxia observed with fentanyl and other MOR agonists.
Why DFNZ Avoids the Pitfalls of Traditional Opioids
The safety profile of DFNZ stems from its unique pharmacokinetic and pharmacodynamic properties. Unlike fentanyl, which enters the brain rapidly and activates μ-opioid receptors with high intrinsic efficacy, DFNZ exhibits slower brain penetration due to PGP-mediated efflux. This delayed onset reduces the risk of respiratory depression, a leading cause of opioid overdose deaths. Additionally, DFNZ’s functional bias toward G-protein signaling over β-arrestin recruitment—unlike morphine, which activates both pathways equally—may contribute to its reduced propensity for addiction and tolerance. β-arrestin signaling is associated with respiratory depression and gastrointestinal side effects, while G-protein signaling is linked to analgesia. By favoring G-protein pathways, DFNZ achieves pain relief without triggering the cascade of adverse effects seen with traditional opioids.
Spatiotemporal Signaling: The Key to DFNZ’s Advantage
Using bioluminescence resonance energy transfer (BRET) assays and cryo-electron microscopy, researchers uncovered DFNZ’s distinct signaling profile. The compound activates G-proteins (Go and Gi1) with high potency and prolonged efficacy, while its recruitment of β-arrestin 1 and β-arrestin 2 is significantly reduced compared to FNZ and DAMGO (a synthetic opioid peptide). This functional bias translates to slower MOR internalization and endosomal trafficking, which may explain DFNZ’s lack of tolerance and withdrawal symptoms. Structural analysis revealed that DFNZ’s binding to the μ-opioid receptor stabilizes an active conformation that preferentially engages G-proteins, providing a molecular basis for its therapeutic advantages.
Abuse Liability: DFNZ Shows Low Reinforcement Potential
One of the most promising findings in the study is DFNZ’s low abuse potential. In intravenous self-administration (IVSA) experiments, rats trained to self-administer heroin significantly reduced their heroin intake when pretreated with DFNZ. However, unlike heroin or morphine, DFNZ did not trigger the characteristic ‘extinction burst’ or reinstatement of drug-seeking behavior—behaviors that signal relapse risk in addiction models. Rats also extinguished lever-pressing for DFNZ-paired cues almost immediately, suggesting minimal reinforcement value. These results align with DFNZ’s weak effect on dopamine neuron activity in the ventral tegmental area, a key region in the brain’s reward circuitry. While FNZ produced a dose-dependent increase in dopamine neuron firing, DFNZ’s effects were modest and delayed, further supporting its reduced addictive potential.
Key Takeaways: What This Discovery Means for Pain Management
- DFNZ, a metabolite of the synthetic opioid FNZ, delivers potent analgesic effects in rodent models with minimal respiratory depression, tolerance, or withdrawal symptoms.
- The compound’s safety stems from its functional bias toward G-protein signaling over β-arrestin recruitment and its limited brain penetration due to P-glycoprotein efflux.
- Unlike traditional opioids like morphine and fentanyl, DFNZ does not induce dopamine-driven reinforcement, suggesting low abuse potential.
- Researchers demonstrated DFNZ’s efficacy in both acute and chronic pain models, including inflammatory pain induced by complete Freund’s adjuvant.
- The discovery highlights the potential of nitazene-derived compounds as a new class of safer painkillers, offering hope amid the ongoing opioid crisis.
Challenges and Next Steps: From Lab to Clinic
While the findings are groundbreaking, translating DFNZ from rodent models to human therapy presents significant challenges. The compound’s preclinical safety profile is promising, but human trials will be necessary to confirm its efficacy and tolerability. Additionally, the nitazene class has a controversial history, and regulatory hurdles could be substantial. The DEA’s recent scheduling of nitazenes as Schedule I substances reflects concerns about their abuse potential, even though DFNZ is structurally distinct and designed for therapeutic use. Researchers are now focusing on optimizing DFNZ’s formulation and delivery methods to enhance its clinical viability. Potential applications include postoperative pain management, chronic pain treatment, and even harm reduction in opioid use disorder, where safer analgesics could reduce reliance on high-potency opioids.
The Broader Implications: A New Chapter in Opioid Pharmacology
The discovery of DFNZ represents a pivotal moment in opioid pharmacology, offering a glimmer of hope in the fight against the opioid crisis. By leveraging the structural versatility of nitazenes while engineering out their toxic properties, researchers have developed a compound that embodies the ideal balance of efficacy and safety. If further studies confirm DFNZ’s potential, it could pave the way for a new generation of μ-opioid receptor agonists—drugs that deliver pain relief without the devastating consequences of addiction and overdose. This paradigm shift underscores the importance of innovative drug design in addressing unmet medical needs, particularly in areas where existing therapies fall short. As the scientific community celebrates this breakthrough, the next phase of research will be critical in determining whether DFNZ can fulfill its promise as a transformative painkiller.
Frequently Asked Questions
Frequently Asked Questions
- Is DFNZ an opioid?
- Yes, DFNZ is a synthetic opioid that selectively activates the μ-opioid receptor, the same target as drugs like morphine and fentanyl. However, its unique structure and functional profile differentiate it from traditional opioids by minimizing respiratory depression and addiction risks.
- How does DFNZ avoid the side effects of other opioids?
- DFNZ achieves this through two primary mechanisms: functional bias toward G-protein signaling over β-arrestin recruitment and limited brain penetration due to P-glycoprotein-mediated efflux. This combination reduces respiratory depression, tolerance, and abuse liability compared to drugs like fentanyl and morphine.
- Has DFNZ been tested in humans?
- As of this publication, DFNZ has only been studied in rodent models. While the results are promising, human clinical trials will be required to confirm its safety and efficacy in people. Researchers are currently preparing for pre-clinical studies to assess its viability for human use.