For nearly a decade, medical guidelines lagged behind scientific evidence—until this week, when cardiology organizations finally updated their lipid panel recommendations to include lipoprotein(a) and apolipoprotein B. Yet even as these long-overdue changes take hold, another critical biomarker, clonal hematopoiesis of indeterminate potential (CHIP), remains sidelined despite overwhelming data linking it to heightened risks for cardiovascular disease, cancer, blood clots, and chronic inflammation. Discovered in 2014, CHIP—an age-related mutation in blood stem cells—now demands urgent clinical attention, with experts arguing that routine screening could save lives by enabling early intervention. Nearly 40% of adults over 90 harbor CHIP mutations, and recent studies reveal its role not just as a risk marker but as a potential driver of some of humanity’s deadliest diseases.
What Is CHIP? The Hidden Genetic Mutation Behind Aging and Disease
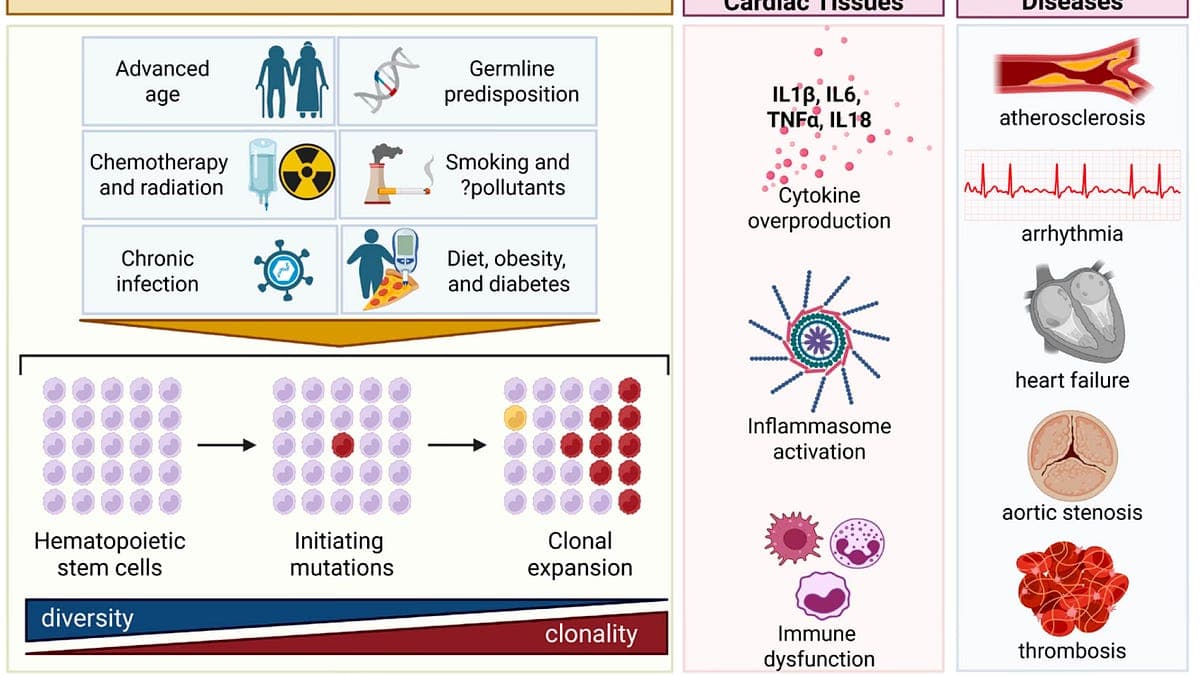
Clonal hematopoiesis of indeterminate potential (CHIP) is a genetic anomaly where a blood stem cell acquires mutations that give it a survival advantage, leading to the proliferation of a mutated clone. While these mutations don’t always cause disease directly, they create a state of chronic inflammation and immune dysfunction—termed "inflammaging" and "immunosenescence"—that accelerates aging and fosters conditions like atherosclerosis, heart attacks, and certain cancers. The marker for CHIP is straightforward: a variant allele frequency (VAF) of 2% or higher in white blood cells, meaning at least 2% of an individual’s immune cells carry the mutation.
The Genetic Landscape of CHIP: Which Mutations Matter Most
CHIP mutations are not random. The most common, accounting for nearly half of all cases, occur in the DNMT3A gene, which regulates DNA methylation. TET2 mutations—responsible for roughly 22% of CHIP cases—are the second most frequent, followed by ASXL1 (11%) and a cluster of others like JAK2, TP53, and SF3B1. These epigenetic mutations disrupt normal cellular function, turning blood stem cells into silent drivers of systemic inflammation. For example, TET2 mutations have been linked to both accelerated aortic aneurysm growth and enhanced immune responses in cancer patients undergoing immunotherapy. "The same mutation can have diametrically opposed effects depending on the tissue context," explains Dr. Eric Topol, founder and director of the Scripps Research Translational Institute, in his widely cited Substack newsletter *Ground Truths*.
How CHIP Risk Escalates with Age and Inflammation
CHIP is strongly age-dependent, with prevalence doubling every decade after age 50. Only 2% of adults aged 50–69 have CHIP, but that jumps to 10% in those 70–79, 20% in the 80–89 bracket, and a staggering 39% in people over 90. This surge coincides with the body’s natural decline in immune function and increased systemic inflammation. The American Heart Association’s 2024 scientific statement on CHIP noted: "Clonal hematopoiesis is increasingly recognized as a potential driver of cardiovascular disease, particularly in older adults where inflammaging and immunosenescence create a fertile ground for disease progression." In patients with solid tumors, CHIP is detected in over 25% of cases—nearly triple the rate seen in healthy age-matched controls—highlighting its role in both cancer susceptibility and treatment response.
The Staggering Health Risks Linked to CHIP
The hazards of CHIP extend far beyond aging. Research published in *The New England Journal of Medicine* and *Circulation* has established that CHIP increases the risk of cardiovascular events by up to 80%—a hazard ratio of 1.8—comparable to the impact of smoking or uncontrolled high cholesterol. For context, hypertension and a total cholesterol level above 200 mg/dL each carry a hazard ratio of 1.4. CHIP’s proinflammatory state also elevates risks for blood clots, liver disease, and multiple cancers, including leukemia and solid tumors like lung and colorectal cancer. "CHIP isn’t just a bystander; it’s an active participant in disease pathogenesis," says Topol. "We’re seeing causal links in both human studies and animal models, where blocking CHIP-related inflammation reduces plaque buildup and tumor progression."
CHIP and Cardiovascular Disease: The Invisible Threat to Heart Health
Atherosclerosis, the buildup of plaque in arteries, is exacerbated by CHIP-driven inflammation. Macrophages—immune cells that engulf cholesterol—become hyperactive in the presence of CHIP mutations like TET2, accelerating plaque formation and increasing the likelihood of heart attacks or strokes. A 2023 study in *Nature Cardiovascular Research* found that individuals with CHIP had a 50% higher incidence of major adverse cardiac events, even after adjusting for traditional risk factors like age, smoking, and diabetes. The worst offenders? Mutations in JAK2, which are associated with a fourfold increased risk of pulmonary embolism, and DNMT3A, which has been linked to early-onset cardiovascular decline.
CHIP in Cancer: A Double-Edged Sword in Treatment and Prognosis
CHIP’s relationship with cancer is complex. In some cases, it worsens outcomes: patients with CHIP undergoing chemotherapy or immunotherapy face higher risks of complications and reduced treatment efficacy. However, certain CHIP mutations—particularly in TET2—can enhance the immune system’s ability to target tumors. A pivotal 2022 study in *Cell* demonstrated that TET2-mutated CHIP improved responses to checkpoint inhibitors in non-small cell lung cancer and colorectal cancer by boosting interferon signaling and T-cell activation. Conversely, in acute myeloid leukemia, CHIP mutations often precede disease onset, serving as early warning signs. "CHIP is a double-edged sword," notes Dr. Lucy Godley, a hematologist at the University of Chicago. "While it can arm the immune system against some cancers, it also primes the ground for others."
Can CHIP Be Treated? Emerging Therapies Offer Hope
Until recently, CHIP was considered untreatable—a silent genetic quirk with no targeted interventions. That paradigm is shifting, thanks to a wave of clinical trials and repurposed drugs. The most compelling evidence comes from low-dose colchicine, an anti-inflammatory medication that reduced CHIP burden by 30% in a randomized placebo-controlled trial. Originally used for gout and pericarditis, colchicine’s ability to inhibit the NLRP3 inflammasome—an immune pathway hyperactive in CHIP—has made it a frontrunner for CHIP management. "Colchicine is cheap, accessible, and well-tolerated," says Topol. "At less than $1 per day, it’s a game-changer for high-risk patients."
Other Promising Interventions: From Metformin to GLP-1 Drugs
Beyond colchicine, several other drugs show potential in targeting CHIP-related inflammation or preventing its progression. Metformin, the diabetes medication, has been linked to reduced CHIP burden in DNMT3A carriers, possibly through its effects on cellular metabolism. Vitamin C may inhibit TET2-mutated CHIP by modulating epigenetic pathways, while interleukin inhibitors like canakinumab (which targets IL-1β) have shown promise in reducing cardiovascular events in high-risk populations. Even GLP-1 drugs, such as semaglutide, are being investigated for their anti-inflammatory properties in CHIP. "The sheer variety of potential treatments underscores how urgently we need clinical trials focused specifically on CHIP," says Dr. Benjamin Ebert, chair of medical oncology at Dana-Farber Cancer Institute. "We’re entering an era where precision medicine could finally catch up to this discovery."
Why Isn’t CHIP Testing Routine? Barriers to Widespread Adoption
Despite the mounting evidence, CHIP testing remains a rarity in clinical practice. The primary obstacle? Cost and complexity. While whole-genome sequencing can detect CHIP, the gold standard is a targeted panel of ~100 genes sequenced at 500x depth—an approach that costs upwards of $950 at labs like Lucence. "This is untenable for most patients," argues Topol. "With sequencing costs plummeting over the past decade, we should be able to offer CHIP testing for under $100." Another hurdle is clinical inertia. Professional societies, including the American Heart Association and American Society of Hematology, have yet to endorse routine CHIP screening, despite their recent acknowledgment of its cardiovascular risks. "It took eight years for guidelines to catch up on lipoprotein(a)—we can’t afford another decade of delay," warns Topol.
The Psychological and Ethical Dilemmas of CHIP Screening
For patients, a positive CHIP result could spark anxiety, particularly for those already at high risk for cancer or heart disease. The fear of false reassurance—or the opposite, unnecessary stress—complicates the push for widespread testing. However, proponents argue that the benefits of early detection outweigh the risks. "Knowing your CHIP status enables targeted prevention," says Dr. Jennifer Kwan of Yale Medicine, which runs a hybrid cardio-oncology CHIP clinic. "For someone with a TET2 mutation, low-dose colchicine or closer cardiovascular monitoring could be lifesaving." Some experts even suggest rebranding CHIP from "indeterminate potential" to "important potential," emphasizing its actionable nature. "The name itself undermines its clinical relevance," Topol argues. "We’re talking about a biomarker with clearer causality than many tests companies like Function Health sell for hundreds of dollars."
The Future of CHIP: Clinics, Trials, and Personalized Medicine
The first dedicated CHIP clinics—at the University of Chicago, Cleveland Clinic, and Yale Medicine—offer a glimpse into the future of personalized medicine. These centers combine cardiology, oncology, and hematology to manage patients with high CHIP burdens, employing risk stratification tools like the newly proposed CHIP risk score, which partitions patients into low-, medium-, and high-risk categories based on mutation type and clone size. Clinical trials are also ramping up, including a phase 2 study of selnoflast, an NLRP3 inflammasome inhibitor, and trials exploring metformin’s potential in DNMT3A-driven CHIP. "The next five years will determine whether CHIP screening becomes standard of care," says Ebert. "If we can prove that intervening early saves lives, there’s no reason this shouldn’t be as routine as cholesterol testing."
Key Takeaways: What You Need to Know About CHIP
- CHIP is an age-related mutation in blood stem cells that increases with age, affecting nearly 40% of people over 90.
- CHIP independently raises the risk of cardiovascular disease, cancer, blood clots, and chronic inflammation, with some mutations (like TET2) having dual roles in disease progression and immune response.
- New treatments, including low-dose colchicine, metformin, and interleukin inhibitors, show promise in reducing CHIP-related risks, but routine testing remains unavailable due to cost and clinical inertia.
- CHIP clinics at leading medical centers are pioneering personalized care, but broader adoption hinges on updated guidelines and affordable testing.
- Rebranding CHIP from "indeterminate" to "important potential" could accelerate its clinical acceptance and public understanding.
Frequently Asked Questions About CHIP
Frequently Asked Questions
- How common is CHIP, and who should be tested?
- CHIP affects about 2% of people aged 50–69, 10% of those 70–79, 20% of 80–89-year-olds, and 39% of people over 90. While routine testing isn’t yet recommended, experts suggest considering it for adults over 65 with a family history of heart disease, cancer, or blood clots, or those with unexplained inflammation.
- What does a positive CHIP test mean for my health?
- A positive CHIP test indicates an elevated risk for cardiovascular disease, certain cancers, and blood clots, but it doesn’t guarantee you’ll develop these conditions. The risk varies by mutation type—for example, JAK2 mutations are strongly linked to clotting, while TET2 mutations may affect cancer treatment responses.
- Are there proven treatments to reduce CHIP-related risks?
- Low-dose colchicine has the strongest evidence for reducing CHIP-driven inflammation and cardiovascular risks. Other potential options include metformin (for DNMT3A-driven CHIP) and interleukin inhibitors, but these are still being studied in clinical trials. Lifestyle changes like exercise and a Mediterranean diet may also help mitigate inflammation.