A team of researchers has developed a groundbreaking machine-learning system that can predict insulin resistance—a key precursor to type 2 diabetes—using data from consumer smartwatches and routine blood tests. The model, described in a new study published in Nature, analyzes heart rate variability, activity levels, sleep patterns, and blood biomarkers to flag individuals at high risk years before traditional diagnostic methods can detect the condition. With 537 million adults worldwide already living with diabetes—a number projected to swell to 643 million by 2030—the discovery offers a desperately needed tool for early intervention, potentially reversing the trajectory of one of the world’s fastest-growing chronic diseases.
- The AI model uses smartwatch data (heart rate, steps, HRV) and blood biomarkers to predict insulin resistance with 80% accuracy
- Detects insulin resistance before blood sugar abnormalities appear, enabling early lifestyle interventions
- Study analyzed 1,165 adults using Fitbit/Pixel Watch data and clinical lab results to train the predictive model
- Foundation model approach captures complex physiological patterns missed by traditional screening methods
- Could reduce reliance on expensive hyperinsulinaemic clamp tests currently used for definitive diagnosis
Why Insulin Resistance Detection Matters Before Diabetes Develops
Insulin resistance—the condition in which the body’s cells become less responsive to insulin—affects an estimated 20-40% of adults globally and precedes type 2 diabetes by 10-15 years in most cases. When pancreatic beta-cells can no longer produce enough insulin to compensate for this resistance, blood glucose levels rise, eventually leading to prediabetes and full-blown diabetes. Unlike type 1 diabetes (an autoimmune disorder), type 2 diabetes is primarily driven by lifestyle factors including obesity, physical inactivity, and poor diet. The condition silently progresses for years before symptoms appear, making early detection critical. While current screening methods like fasting glucose tests or HbA1c measurements often miss early-stage resistance, this new model identifies metabolic dysfunction at a stage when interventions—such as weight loss, exercise, or medication—can still reverse the disease process.
The Hidden Costs of Late-Stage Diabetes Detection
The economic burden of type 2 diabetes is staggering, costing the U.S. healthcare system an estimated $413 billion annually in direct medical expenses and lost productivity. Once diabetes develops, patients face irreversible complications including cardiovascular disease, kidney failure, nerve damage, and vision loss. Early detection of insulin resistance could prevent up to 70% of diabetes cases, according to the Centers for Disease Control and Prevention. Yet 80% of prediabetic individuals remain undiagnosed. This new screening approach leverages data already being collected by millions of smartwatch users, potentially making preventive care accessible to populations that lack regular access to healthcare providers.
How the AI Model Predicts Insulin Resistance Before Symptoms Appear
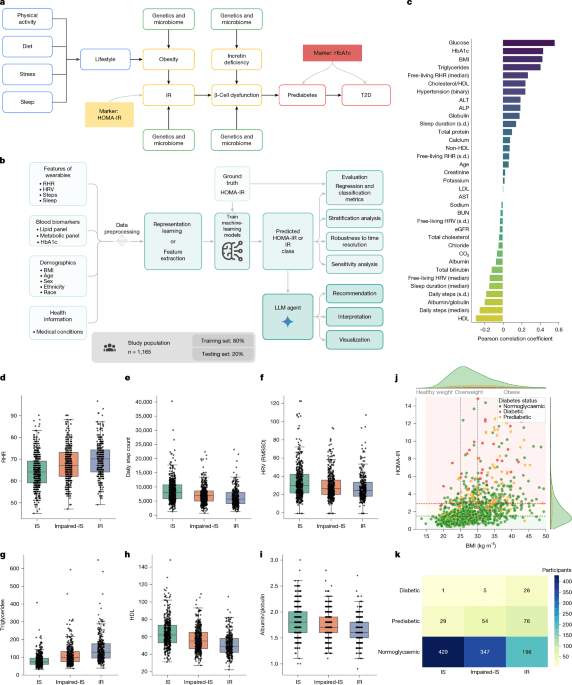
The research team, led by scientists at Google Health and multiple academic institutions, developed what they call a multimodal machine-learning framework that integrates three key data streams: continuous physiological signals from wearable devices, routine blood biomarkers, and basic demographic information. Their Wearables for Metabolic Health (WEAR-ME) study enrolled 1,165 U.S. adults who wore Fitbit devices or Google Pixel watches while providing blood samples through Quest Diagnostics.
The Three Pillars of Prediction: Wearables, Blood Tests, and Demographics
At the heart of the model is a deep learning system that processes high-resolution wearable data—capturing metrics like resting heart rate (RHR), heart rate variability (HRV), step count, and sleep duration at one-minute intervals. These physiological signals reveal metabolic dysfunction through patterns invisible to traditional screening. For example, individuals with insulin resistance tend to have higher resting heart rates and lower HRV, indicators of autonomic nervous system dysfunction linked to metabolic disorders. The model couples this rich time-series data with routinely available blood biomarkers including fasting glucose, triglycerides, HDL cholesterol, and HbA1c.
- Resting heart rate positively correlates with insulin resistance (r=0.27, P<0.001)
- Heart rate variability shows strong inverse correlation with resistance (r=-0.29, P<0.001)
- Daily step count negatively correlates with insulin resistance (r=-0.19, P<0.001)
- Fasting glucose alone doubled the model’s predictive accuracy when added to wearable data
- Combined model achieved 80% AUROC (area under receiver operating characteristic curve)
The Breakthrough: Foundation Models That Learn from Raw Sensor Data
What sets this research apart is the use of a wearable foundation model—a type of AI system pretrained on massive amounts of unlabeled wearable data to learn robust representations of complex physiological patterns. Unlike previous approaches that relied on simple averages of heart rate or step counts, this foundation model processes the raw minute-by-minute signals to capture subtle variations in activity, sleep, and cardiovascular responses that indicate metabolic dysfunction. When researchers fine-tuned this model with demographic and blood biomarker data, it achieved unprecedented predictive power. The foundation model approach increased the importance of wearable data in predictions from 43% to 82%, demonstrating its ability to extract meaningful patterns from the vast streams of sensor information.
Our results show that the detailed embedding of the foundation model captures the complex dynamics and interplay of daily activity, sleep and physiological rhythms that are crucial to insulin resistance but missed by simple aggregates.
From Research to Reality: How This System Could Transform Healthcare
Currently, the gold standard for diagnosing insulin resistance—the hyperinsulinaemic euglycaemic clamp—is rarely used outside research settings due to its complexity, cost ($1,000-$2,000 per test), and time requirements (3-4 hours per patient). The homeostatic model assessment (HOMA-IR), calculated from fasting insulin and glucose levels, serves as a more practical alternative but still requires clinical lab visits and insulin testing. However, insulin assays aren’t typically included in routine annual exams, and home testing kits remain unavailable. This creates a diagnostic gap where millions of people with early metabolic dysfunction remain undetected until irreversible damage occurs.
A Screening Tool That Prioritizes High-Risk Individuals for Clinical Testing
The researchers envision their model serving as a first-line screening tool that identifies individuals most likely to benefit from confirmatory clinical testing. With 26% of U.S. adults now owning smartwatches and 15% undergoing annual exams with blood biomarker assessment, the infrastructure for widespread implementation already exists. Positive AI screenings would prompt targeted insulin and glucose testing to calculate precise HOMA-IR values, enabling clinicians to intervene before diabetes develops. This approach could dramatically reduce healthcare costs while improving outcomes—particularly for populations with limited access to preventive care.
The AI Doctor is In: How Large Language Models Could Explain Your Risk
Beyond prediction, the research team developed an AI-powered health assistant called the Insulin Resistance Literacy and Understanding Agent (IR agent) that interprets model results in plain language. Built on Google’s Gemini 2.0 Flash, this system combines the predictive model’s output with user health data to provide personalized explanations and lifestyle recommendations. In blinded evaluations by five board-certified endocrinologists, the IR agent’s responses were preferred 73-92% of the time over a standard AI model without metabolic risk information. The system achieved 79% factual accuracy and 96% safety ratings, demonstrating its potential as a patient-facing tool for metabolic health education.
What Users Could Ask Their AI Health Assistant
The IR agent demonstrates how future health management systems might work in practice. A user concerned about their risk might ask: 'Doctor, my smartwatch shows my resting heart rate is 78 beats per minute. Should I be worried about diabetes?' The agent would analyze this physiological data alongside blood biomarkers and demographic factors to respond with context-specific guidance, such as: 'Your resting heart rate of 78 bpm combined with your fasting glucose of 95 mg/dL suggests early insulin resistance. Consider discussing a Mediterranean diet and 150 minutes of moderate exercise weekly with your physician.' Such personalized guidance could empower individuals to take preventive action years before traditional symptoms emerge.
Limitations and Future Directions: What’s Next for This Technology
While promising, the research faces several challenges that must be addressed before widespread clinical implementation. The study cohort, though diverse in age and gender, overrepresented health-conscious individuals willing to participate in remote monitoring studies. Only 25% of enrolled participants provided complete data, potentially skewing results toward those already engaged in health tracking. Additionally, the model’s performance varied across BMI categories, with underweight individuals showing different predictive patterns than those with obesity. Future research should incorporate genetic data, microbiome analysis, and more diverse wearable brands to improve generalizability. The researchers also acknowledge limitations in using HOMA-IR as ground truth, noting that insulin measurements can vary 23.5% between laboratory tests.
The Bigger Picture: How This Could Change Chronic Disease Management
This breakthrough represents a paradigm shift in chronic disease prevention, moving from reactive treatment to proactive, data-driven health optimization. The same approach could be adapted to predict risks for cardiovascular disease, metabolic dysfunction-associated steatotic liver disease (MASLD), and other obesity-related conditions. Insurance companies and healthcare systems are already exploring how such models could reduce long-term costs by identifying high-risk individuals before expensive complications develop. The researchers note particular promise in using insulin resistance detection to guide prescriptions of GLP-1 and GIP receptor agonists, medications currently prescribed based primarily on BMI thresholds that may not accurately reflect metabolic need.
Key Takeaways: What Consumers and Healthcare Providers Should Know
- This AI model can predict insulin resistance years before traditional tests detect abnormalities
- Smartwatch data combined with routine blood tests achieves 80% accuracy in identifying high-risk individuals
- Early detection enables lifestyle changes or medications that can prevent or reverse type 2 diabetes
- The system uses data already collected by millions of wearable devices, making it scalable and accessible
- A companion AI assistant can explain risk levels and provide personalized recommendations
Frequently Asked Questions
Frequently Asked Questions
- How accurate is this AI model compared to traditional diabetes screening?
- The model achieves 80% accuracy in predicting insulin resistance, significantly outperforming traditional screening methods that often miss early-stage resistance. When combined with fasting glucose, it reaches 87% accuracy—comparable to some clinical tests but available through consumer devices.
- Do I need special equipment or can any smartwatch work?
- The researchers used Fitbit and Pixel Watch devices, but the model was designed to work with standard wearable features like heart rate monitoring and step counting. Lower-cost trackers with these capabilities should be compatible, though data quality may vary between brands.
- What would I need to do if the AI flagged me as high-risk?
- If flagged as high-risk, you should discuss confirmatory testing with your healthcare provider. This typically involves a fasting glucose test and insulin measurement to calculate your HOMA-IR score. Your doctor may recommend lifestyle changes, medications, or further monitoring based on your individual risk profile.