Multiple sclerosis (MS) has long been recognized for its devastating impact on the central nervous system, but a groundbreaking study published in Nature now reveals the precise cellular mechanisms driving selective neurodegeneration in the disease. Researchers have discovered that DNA damage accumulates in specific neurons located in the upper layers of the cerebral cortex, leading to their progressive loss during neuroinflammatory episodes. These neurons, marked by the CUX2 protein, are uniquely vulnerable to damage caused by inflammation, oxidative stress, and inadequate DNA repair mechanisms. The findings, based on human tissue analysis and mouse models, provide critical insights into why MS patients experience cortical thinning and cognitive decline, while also identifying potential therapeutic targets to protect these vulnerable neurons.
Why Upper-Layer Neurons Are Particularly Vulnerable in Multiple Sclerosis
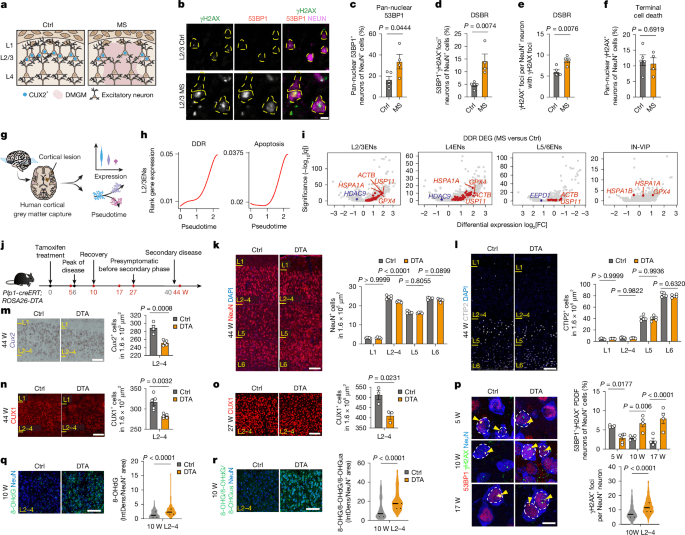
Multiple sclerosis is characterized by immune system attacks on the myelin sheaths that protect nerve fibers in the brain and spinal cord. While early treatments focused on reducing immune attacks, many patients still experience progressive disability due to ongoing neurodegeneration. Researchers have now identified a specific population of neurons—those in cortical layers 2 and 3 that express the CUX2 protein—as being particularly susceptible to damage. Unlike deeper-layer neurons, these cells fail to adequately repair DNA damage caused by inflammation and oxidative stress. The study found that even when the immune system is suppressed, these neurons continue to degenerate due to intrinsic vulnerabilities in their DNA repair machinery.
The Role of DNA Damage in Neurodegeneration
Every cell in the body accumulates DNA damage throughout life, but neurons are particularly vulnerable because they do not divide and repair much more slowly. In MS, the accumulation of DNA damage is accelerated by neuroinflammation, which generates reactive oxygen species and other toxic byproducts. The study’s authors found that neurons in MS patients’ brains showed elevated markers of DNA damage, including 53BP1 and γH2AX foci, which indicate double-stranded DNA breaks and other forms of damage. These markers were particularly concentrated in CUX2-expressing neurons in layers 2 and 3 of the cortex.
The DNA damage response (DDR) system is supposed to repair these breaks, but the study found that CUX2 neurons in MS brains had an impaired DDR. Instead of repairing damage, these neurons showed signs of transcriptional dysregulation and eventual cell death. This was not the case for deeper-layer neurons or other cell types in the cortex, which maintained relatively normal DNA repair functions.
How Neuroinflammation Accelerates Neuronal Damage
Neuroinflammation is a hallmark of MS, driven by immune cells infiltrating the brain and releasing toxic molecules like interferon-gamma (IFNγ). The study found that IFNγ directly damages neurons by increasing oxidative stress, which in turn causes DNA damage. In human neuron models, IFNγ exposure led to a 15% reduction in cell viability, accompanied by elevated DNA damage markers. This effect was mitigated by antioxidants like N-acetylcysteine (NAC) and Mito-TEMPO, which neutralize reactive oxygen species.
The Timing of Neuronal Loss in MS Progression
The study used mouse models of MS to track the progression of neuronal damage over time. In one model, widespread oligodendrocyte death led to a delayed but progressive loss of CUX2 neurons in layers 2 and 3, even though inflammation peaked early. This delayed neurodegeneration suggests that initial immune attacks prime neurons for future damage, which only becomes apparent months later when repair mechanisms fail.
In another model, astrocytes were engineered to produce IFNγ in the cortex. This led to an 18% reduction in CUX2 neurons by 52 weeks, without significant demyelination. This finding underscores the direct toxic effects of neuroinflammation on these vulnerable neurons, independent of myelin loss.
The Critical Role of CUX2 and ATF4 in DNA Repair
The study identified two key proteins—CUX2 and ATF4—that play crucial roles in repairing DNA damage in neurons. CUX2, a transcription factor expressed predominantly in layers 2 and 3 neurons, was found to activate the RPA3 gene, which is essential for repairing double-stranded DNA breaks. When CUX2 function was disrupted, neurons showed increased DNA damage and vulnerability to neuroinflammatory stress. Similarly, ATF4 was shown to enhance DNA repair by promoting non-homologous end joining (NHEJ), a critical repair pathway for neurons.
In experiments using human neuron models, overexpression of CUX2 or ATF4 significantly reduced DNA damage and improved cell survival after exposure to oxidative stress. The findings suggest that boosting these proteins’ activity could protect neurons from damage in conditions like MS.
Implications for Multiple Sclerosis Treatment and Research
The discovery of CUX2 neurons’ vulnerability to DNA damage and their impaired repair mechanisms offers new avenues for MS treatment. Current therapies focus on suppressing the immune system, but this study suggests that combining immune therapies with neuroprotective strategies could yield better outcomes. Potential approaches include antioxidants to reduce oxidative stress, drugs that enhance DNA repair, or therapies that block IFNγ signaling to prevent neuronal damage.
The findings also highlight the importance of early intervention. Since DNA damage accumulates over time, therapies that bolster DNA repair or reduce oxidative stress may be most effective when started early in the disease course. This could help prevent the cortical thinning and cognitive decline that are hallmarks of progressive MS.
Key Takeaways About DNA Damage and Neuronal Loss in Multiple Sclerosis
- DNA damage accumulates in specific neurons (CUX2-expressing neurons in layers 2 and 3) in multiple sclerosis brains, leading to their selective loss.
- Neuroinflammation, particularly interferon-gamma (IFNγ), directly damages neurons by increasing oxidative stress and impairing DNA repair mechanisms.
- CUX2 and ATF4 are critical proteins that repair DNA damage in neurons. Boosting their function may protect neurons from degeneration.
- Antioxidants like NAC and Mito-TEMPO can mitigate DNA damage caused by neuroinflammation, offering potential therapeutic benefits.
- Early intervention with neuroprotective strategies may prevent cortical thinning and cognitive decline in MS patients.
Frequently Asked Questions About Multiple Sclerosis and Neuronal DNA Damage
Frequently Asked Questions
- What causes DNA damage in multiple sclerosis?
- DNA damage in MS is caused by a combination of intrinsic factors like metabolic stress and external factors such as neuroinflammation, oxidative stress, and immune system attacks on the brain. These stressors generate reactive oxygen species and other toxic molecules that damage DNA.
- Why are CUX2 neurons particularly vulnerable in MS?
- CUX2 neurons in layers 2 and 3 of the cortex are vulnerable because they have impaired DNA repair mechanisms. These neurons rely on proteins like CUX2 and ATF4 to repair damage, but inflammation disrupts these pathways, leading to progressive cell loss.
- Can antioxidants help protect neurons in MS?
- Yes, antioxidants like N-acetylcysteine (NAC) and Mito-TEMPO have shown promise in reducing oxidative stress and DNA damage in neurons. These therapies may help protect vulnerable neurons and slow disease progression.