In a groundbreaking advance that blurs the line between biology and silicon, researchers at the University of Massachusetts Amherst have engineered the world’s first artificial neuron capable of operating at the same electrical voltage as living human brain cells. The breakthrough, published in the journal Nature Electronics, represents a critical step toward seamless integration between electronic devices and biological systems—potentially enabling everything from ultra-precise brain-machine interfaces to implantable health monitors that communicate using the body’s own electrical language.
Why This Artificial Neuron Changes Everything for Bioelectronics
For decades, engineers have struggled to create artificial neurons that can directly interface with biological tissues. The core obstacle has been voltage mismatch: while natural neurons fire electrical spikes between 70 and 130 millivolts (mV), most artificial neurons required 500 mV or more to operate. This disparity created a fundamental barrier. Stronger electrical signals from artificial systems could either overpower delicate biological activity or require complex amplification hardware—adding bulk, energy demands, and latency to devices like pacemakers, cochlear implants, or brain-computer interfaces. The UMass Amherst team, led by Jun Yao, has now closed this gap by developing a bioinspired neuron that operates at ~60–100 mV, matching the natural range of human neurons. 'Previous versions of artificial neurons used 10 times more voltage and 100 times more power than the one we have created,' Yao said. 'That gap wasn’t just inefficient—it made direct contact nearly impossible.'
The Science Behind the Breakthrough: Memristors and Bacterial Nanowires
At the heart of this innovation is a memristor—a nanoscale electronic component whose resistance changes in response to electrical current. Unlike traditional transistors, memristors 'remember' their state even after power is removed, mimicking the adaptive behavior of biological synapses. The UMass team engineered their memristor using bacterial nanowires derived from Geobacter sulfurreducens, a soil-dwelling microbe already renowned for its ability to conduct electricity.
These nanowires, just a few nanometers in diameter, were strategically integrated into the memristor to tune its electrical behavior. When tested, the device exhibited a critical threshold: it flipped on at approximately 60 mV and 1.7 nanoamps of current, then automatically reset—replicating the transient 'spike' characteristic of real neurons. This self-resetting mechanism allowed the artificial neuron to mimic not only the shape of a neural signal but also its timing and energy efficiency. 'The on-off behavior isn’t arbitrary,' explained Yao. 'It mirrors the refractory period in biological neurons, where a cell must reset before firing again.'
From Lab Circuit to Living Tissue: Proving Real-World Compatibility
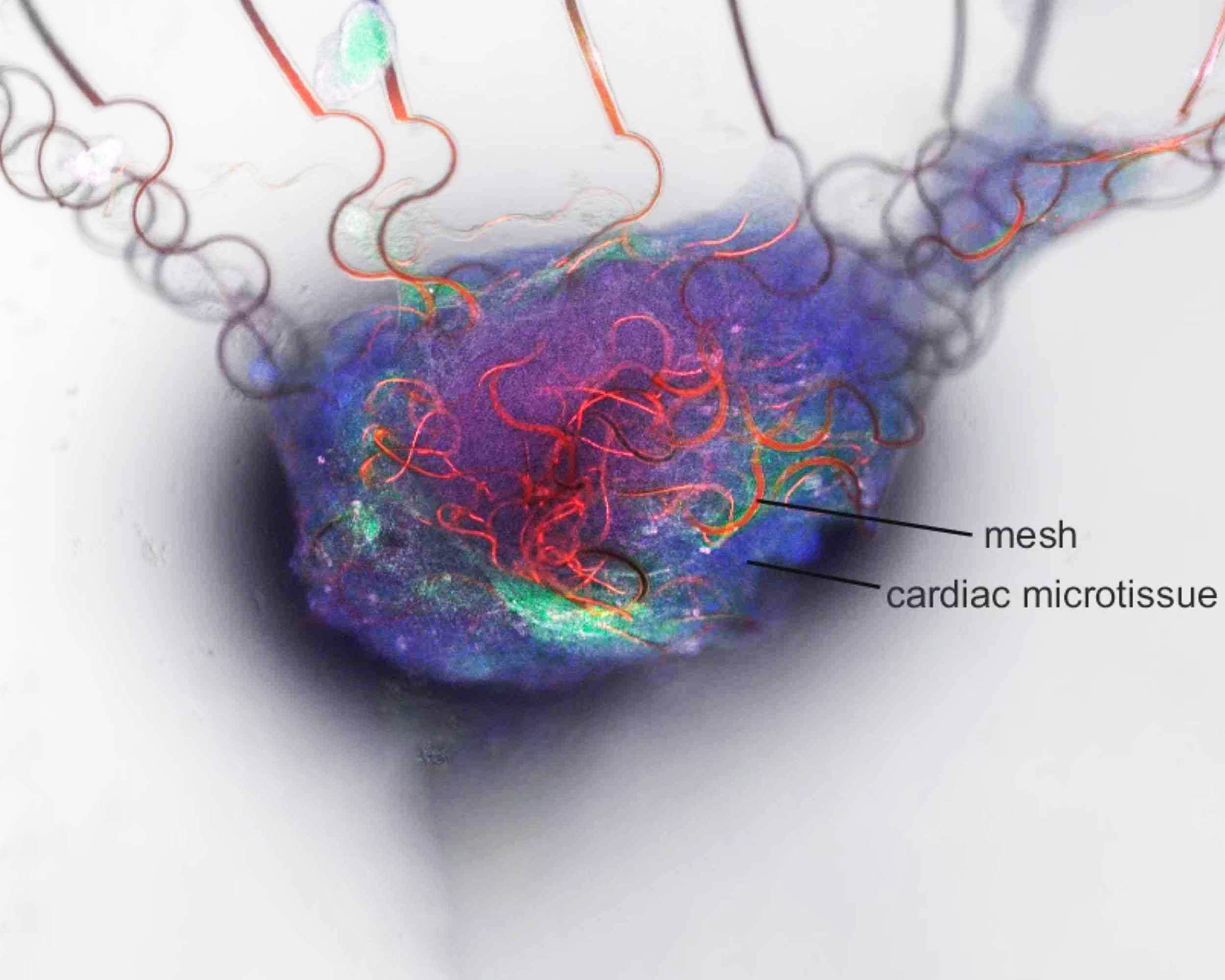
While the artificial neuron’s performance in a controlled circuit was impressive, the team sought to validate its function in a living biological system. They chose cardiomyocytes—heart muscle cells that generate rhythmic electrical impulses as they contract. Using a soft graphene mesh embedded with sensors, the researchers cultured heart tissue directly onto the device. Under normal conditions, the artificial neuron remained silent, but when a drug was introduced to accelerate the heart cells’ rhythm, the circuit fired in response—demonstrating real-time electrical communication.
This experiment did not replicate a human brain interface, but it proved that the artificial neuron could 'listen' to biological signals, process them within its own low-voltage framework, and respond appropriately. Importantly, the device did so without external amplification—a common bottleneck in today’s wearable and implantable sensors. 'That intermediate step of amplification increases both power consumption and circuit complexity,' Yao noted. 'Our neurons eliminate that need entirely, operating at the raw signal level.'
How Chemical Signals Bring Intelligence to Artificial Neurons
The UMass Amherst team went beyond replicating electrical patterns by embedding chemical sensing capabilities into the artificial neuron. The human brain doesn’t rely solely on electrical impulses; it’s profoundly influenced by neurotransmitters like dopamine and sodium ions that modulate neural activity. The researchers incorporated graphene-based sensors into the circuit, allowing it to respond to chemical cues in real time. For example, elevated sodium levels sped up the neuron’s reset phase, causing it to fire more frequently—mirroring how biological neurons become more excitable under high salt conditions. 'Neuromodulation is a critical layer of neural computation,' said Yao. 'Ignoring it would be like designing a computer without considering software inputs.'
Dopamine, a key player in reward and motivation circuits, produced a bidirectional effect. At certain concentrations, it increased the neuron’s firing rate, while higher doses dampened activity—a response analogous to how dopamine modulates movement in Parkinson’s disease and addiction. This chemical responsiveness suggests future applications not only in biosensors but also in closed-loop therapeutic devices, such as deep brain stimulators for neurological disorders.
Energy Efficiency: A Fraction of the Cost of Previous Models
Energy consumption has long been a limiting factor in bioelectronic devices. Traditional artificial neurons required milliwatts of power to operate, making long-term implants impractical. The UMass Amherst neuron, by contrast, operates in the nanowatt range—around 100 times more efficient than earlier chemical-based artificial neurons. The simplified circuit, stripped of unnecessary amplification stages, maintained the same spiking energy even when core sensing components were removed. 'We’ve reduced the power budget to a level compatible with body-powered devices,' Yao said. This efficiency could enable future wearable patches or subcutaneous implants that run for months or years on a single charge.
Scalability and Fabrication: Building Toward Real-World Applications
One of the most promising aspects of this technology is its compatibility with existing semiconductor manufacturing processes. The artificial neuron is built using standard silicon-friendly fabrication techniques, meaning it could be mass-produced using infrastructure already in place at chip foundries. 'We’re not inventing new materials or exotic processes,' Yao noted. 'We’re adapting what’s already proven to work in electronics.' This scalability is crucial for translating lab prototypes into medical-grade devices or even consumer-grade health monitors.
Still, significant challenges remain. The current prototype exists on a laboratory bench, not as a commercial product. Long-term stability, biocompatibility, and integration with complex neural networks will require extensive testing—especially in human tissue and over extended periods. Regulatory hurdles for medical devices are substantial, and ethical concerns about brain-machine interfaces persist. But the foundational leap is undeniable: for the first time, an artificial neuron speaks the same electrical language as living cells.
Broader Implications: From Medicine to Machine Intelligence
The implications of this breakthrough extend far beyond medical implants. By enabling direct, low-energy communication between electronics and biology, the technology could accelerate the development of electroceuticals—drugs that work by interfacing with the nervous system. It also opens new avenues in neuromorphic computing, where processors mimic the brain’s architecture to perform complex tasks with minimal power. Imagine smartphones that interface with your nervous system to reduce cognitive load, or prosthetic limbs that move intuitively based on neural feedback. 'This isn’t just about fixing broken systems,' said Yao. 'It’s about creating a new kind of symbiosis between humans and machines.'
Key Takeaways
- Researchers at UMass Amherst developed the first artificial neuron that operates at biological voltage levels (60–100 mV), matching real neurons’ electrical signals.
- The breakthrough relies on memristors tuned with bacterial nanowires and graphene-based chemical sensors, enabling low-energy, direct bioelectronic communication.
- In tests with heart cells, the artificial neuron responded to biological signals without amplification—demonstrating real-time compatibility.
- Chemical modulation via dopamine and sodium shows the neuron can replicate not just electrical but also biochemical neural processing.
- With energy efficiency 100 times greater than prior models and scalable silicon fabrication, the technology is poised for broader applications in medicine and computing.
What’s Next? The Path to Implants and Brain Networks
While this achievement marks a historic milestone, the road to clinical or commercial use is long. The team plans to test the neuron with true nerve tissue, including rodent models, to assess long-term stability and safety. They’re also exploring how arrays of these neurons could form basic neural networks, mimicking the parallel processing of the brain. 'We’re not claiming this is a brain implant yet,' Yao cautioned. 'But we’ve removed the biggest technical barrier: the inability of electronics to speak biology’s language.'
“By speaking in signals that living cells already use, the artificial neuron made efficiency and compatibility part of the same story.” — Jun Yao, University of Massachusetts Amherst
Frequently Asked Questions
Frequently Asked Questions
- Can this artificial neuron be used in brain implants today?
- Not yet. The current prototype is a laboratory device and has not undergone the rigorous safety and biocompatibility testing required for human use. Regulatory approval would be necessary before any clinical application.
- How does this neuron compare to existing brain-computer interfaces like Neuralink?
- Current BCIs like Neuralink use electrodes to record neural activity and transmit data externally. This artificial neuron, however, is designed to both sense biological signals and generate its own electrical responses—potentially enabling two-way communication at the cellular level.
- What are the energy requirements of this artificial neuron?
- The neuron operates in the nanowatt range—about 100 times less power than earlier artificial neurons. This efficiency could allow for long-term, battery-powered implants or even energy-harvesting devices powered by body heat or movement.